Trending...
- Peak 10 Marketing Expands Capabilities and Opens Doors to New Clients
- Maryland Department of Agriculture Accepting 2026 Animal Waste Technology Fund Proposals
- How LIB's Temperature & Humidity Chamber & Walk-in Chamber Warranty Delivered Real Uptime
MUNICH - Marylandian -- OncoBeta® GmbH, a medical device company specialising in innovative epidermal radioisotope therapies, has announced 12-months interim results from its international Phase IV, multi-centre clinical trial evaluating the efficacy and safety of Rhenium-SCT® (Skin Cancer Therapy) in patients with non-melanoma skin cancer (NMSC).
The EPIC-Skin Study (Efficacy of Personalised Irradiation with Rhenium-SCT for the Treatment of Non-Melanoma Skin Cancer) is designed to assess treatment efficacy and safety, as well as key patient-reported outcomes including quality of life, treatment comfort, and cosmetic results.1 The study is based on the clinically validated effect of the beta-emitter rhenium-188 in treating basal cell carcinoma (BCC) and squamous cell carcinoma (SCC).2
Treatment data is available for 184 adult patients (with 254 lesions) across 7 study centres in Australia, Austria, Germany, the United Kingdom, and South Africa. All participants had histologically confirmed stage I or II NMSC. The median patient age was 70.3 years (range 27–95 years), and 53.3% of participants were male. Patients had one (71.2%), two (19.6%), or three (9.2%) lesions treated, which were located on the head & neck (11.8%), trunk (11.8%), lower limbs (11.8%), or upper limbs (9.1%).1
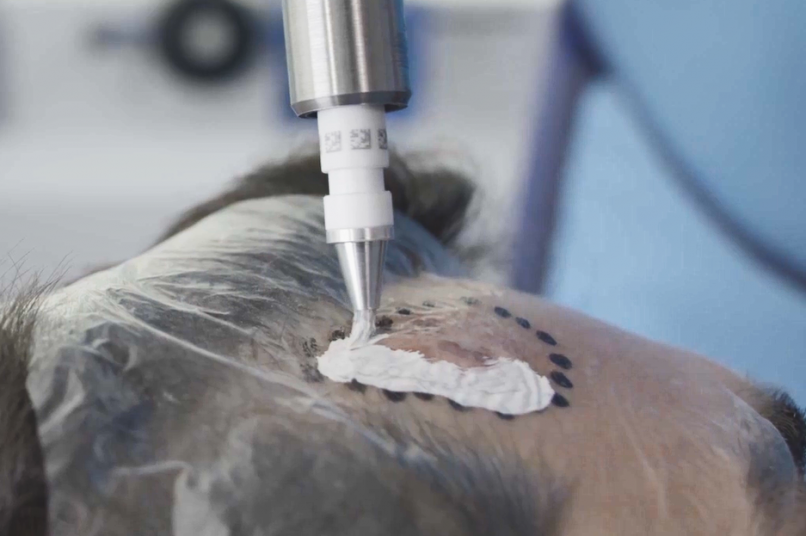
Treatment & Methodology
Rhenium-SCT was administered as a single 50-Gy topical dose of rhenium-188 embedded in a resin applied via adhesive film to the lesion site. Efficacy was assessed using modified RECIST criteria, at 12 months follow-up. Quality of life was assessed using the Skin Cancer Index (SCI) at baseline, 6 months, and 12 months. Treatment comfort was assessed using a patient questionnaire, whilst patient- and clinician-evaluated cosmetic outcomes were determined using a visual analogue scale (VAS) grading of 0-10 (0 = very poor; 10 = no wound detectable). Safety was assessed via treatment-emergent adverse events (TEAEs) and CTCAE (Common Terminology Criteria for Adverse Events) grading.
Key 12-Month Results:1
These findings confirm that Rhenium-SCT®, administered in a single session, delivers sustained high efficacy and improved quality of life, with excellent cosmetic outcomes and minimal adverse effects.
More on Marylandian
"Rhenium-SCT® has consistently demonstrated effectiveness and safety in prior studies," said Dr. Gerhard Dahlhoff, Medical Director at OncoBeta®. "This 12-month interim data further reinforces its value as a non-invasive, targeted treatment option for NMSC, particularly for patients seeking alternatives to surgery due to cosmetic or health-related concerns."
"The 12-month results from the EPIC-Skin Study represent a major milestone for OncoBeta®," added Shannon D. Brown III, CCO OncoBeta & Managing Director Europe. "These results support the efficacy, safety, and patient satisfaction associated with Rhenium-SCT®. These are the three foundational pillars of our patient-first vision, emphasising not only clinical outcomes, but also the patient experience in achieving them."
_
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.3 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.4
The Rhenium-SCT is a painless*, non-invasive‡ therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,5,6 The Rhenium-SCT utilises the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy2 that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.6-8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta® has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting non-melanoma skin cancers and keloid scars. OncoBeta® has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
More on Marylandian
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties, and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta® undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No pain reported during procedure.6
‡No direct skin contact or incisions.6
References
The EPIC-Skin Study (Efficacy of Personalised Irradiation with Rhenium-SCT for the Treatment of Non-Melanoma Skin Cancer) is designed to assess treatment efficacy and safety, as well as key patient-reported outcomes including quality of life, treatment comfort, and cosmetic results.1 The study is based on the clinically validated effect of the beta-emitter rhenium-188 in treating basal cell carcinoma (BCC) and squamous cell carcinoma (SCC).2
Treatment data is available for 184 adult patients (with 254 lesions) across 7 study centres in Australia, Austria, Germany, the United Kingdom, and South Africa. All participants had histologically confirmed stage I or II NMSC. The median patient age was 70.3 years (range 27–95 years), and 53.3% of participants were male. Patients had one (71.2%), two (19.6%), or three (9.2%) lesions treated, which were located on the head & neck (11.8%), trunk (11.8%), lower limbs (11.8%), or upper limbs (9.1%).1
Treatment & Methodology
Rhenium-SCT was administered as a single 50-Gy topical dose of rhenium-188 embedded in a resin applied via adhesive film to the lesion site. Efficacy was assessed using modified RECIST criteria, at 12 months follow-up. Quality of life was assessed using the Skin Cancer Index (SCI) at baseline, 6 months, and 12 months. Treatment comfort was assessed using a patient questionnaire, whilst patient- and clinician-evaluated cosmetic outcomes were determined using a visual analogue scale (VAS) grading of 0-10 (0 = very poor; 10 = no wound detectable). Safety was assessed via treatment-emergent adverse events (TEAEs) and CTCAE (Common Terminology Criteria for Adverse Events) grading.
Key 12-Month Results:1
- Overall response rate: 97.3%
- Complete response rate: 94.1%
- Partial response rate: 3.2%
- Mean improvement in quality of life: +10.55 points from baseline
- Pain/discomfort during treatment: None reported
- Cosmetic outcomes: Favourable results reported by both patients and clinicians
- Most common toxicity (12-month): Grade 1 hypopigmentation (60.4%)
- No CTCAE toxicities above Grade 2 observed at 12 months
These findings confirm that Rhenium-SCT®, administered in a single session, delivers sustained high efficacy and improved quality of life, with excellent cosmetic outcomes and minimal adverse effects.
More on Marylandian
- "Leading From Day One: The Essential Guide for New Supervisors" Draws from 25+ Years of International Management Experience
- New Slotozilla Project Explores What Happens When the World Goes Silent
- IASO Bio to Present Promising Findings on Equecabtagene Autoleucel for Multiple Sclerosis at both ANA and ECTRIMS 2025
- The Two Faces of Charles D. Braun: How the Novel, Posthumously Yours, Came to Life
- Goodwill Industries of the Chesapeake Celebrates Second Graduating Class from Excel Center, Baltimore's Tuition-Free Adult High School
"Rhenium-SCT® has consistently demonstrated effectiveness and safety in prior studies," said Dr. Gerhard Dahlhoff, Medical Director at OncoBeta®. "This 12-month interim data further reinforces its value as a non-invasive, targeted treatment option for NMSC, particularly for patients seeking alternatives to surgery due to cosmetic or health-related concerns."
"The 12-month results from the EPIC-Skin Study represent a major milestone for OncoBeta®," added Shannon D. Brown III, CCO OncoBeta & Managing Director Europe. "These results support the efficacy, safety, and patient satisfaction associated with Rhenium-SCT®. These are the three foundational pillars of our patient-first vision, emphasising not only clinical outcomes, but also the patient experience in achieving them."
_
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.3 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.4
The Rhenium-SCT is a painless*, non-invasive‡ therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,5,6 The Rhenium-SCT utilises the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy2 that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.6-8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta® has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting non-melanoma skin cancers and keloid scars. OncoBeta® has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
More on Marylandian
- Counseling Center of New Smyrna Beach Expands Affordable Mental Health Services for Volusia County
- Maryland Agricultural Land Preservation Foundation (MALPH) Board of Trustees Meeting
- Athena Forge (ATFG) Introduces Advanced Token for Technology-Driven Financial Ecosystem
- Albuquerque's Z-CoiL Footwear Brings All-American Family Business Story to Shark Tank Season Premiere
- NoviSign Sponsoring VARTECH 2025 - the B2B IT channel's #1 event
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties, and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta® undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No pain reported during procedure.6
‡No direct skin contact or incisions.6
References
- Cardaci G, et al. Adv Radiat Oncol. 2025;10(7):101802.
- Cipriani C, et al. J Dermatol Treat. 2022;33(2):969–975. Epub 22 Jul 2020.
- Ciążyńska M, et al. Sci Rep. 2021;11(1):4337.
- Cancer.net. Skin Cancer (Non-Melanoma): Risk Factors and Prevention. February 2022. https://www.cancer.net/cancer-types/skin-cancer-non-melanoma/risk-factors-and-prevention (accessed June 2025).
- Tietze JK, et al. Clin Nuc Med. 2023;48(10):869–876.
- Castellucci P, et al. Eur J Nucl Med Mol Imaging. 2021;48(5):1511–1521.
- Cipriani C, et al. Int J Nucl Med. 2017;114–112.
- Sedda AF, et al. Clin Exp Dermatol. 2008;33(6):745–749.
Source: OncoBeta GmbH
0 Comments
Latest on Marylandian
- Sheets.Market Brings Professional Financial Model Templates to Entrepreneurs and Startups
- Webinar Announcement: Investing in the European Defense Sector—How the New Era of Uncertainty Is Redefining Investment Strategies
- AEVIGRA (AEIA) Analysis Reveals $350 Billion Counterfeit Market Driving Luxury Sector Toward Blockchain Authentication
- RUNA Brings Celtic Spirit and American Roots to New Spire Stages
- Her Magic Mushroom Memoir Launches as a Binge-Worthy Novel-to-Podcast Experience
- Century Fasteners de Mexico Hires Saúl Pedraza Gómez as Regional Sales Manager in Mexico
- Georgia Misses the Mark Again on Sports Betting, While Offshore Sites Cash In
- Maryland: Invasive Plant Advisory Committee (IPAC) Meeting Notice
- $40 Price Target for $NRXP in H. C. Wainright Analyst Report on Leader in $3 Billion Suicidal Depression Market with Superior NRX 100 Drug Therapy
- Nashville International Chopin Piano Competition Partners with Crimson Global Academy to Support Excellence in Education
- AHRFD Releases Market Analysis: Cryptocurrency Market's Institutional Transformation Accelerating
- Ubleu Crypto Group Analyzes European Digital Asset Market Opportunities Amid Regulatory Evolution
- NIUFO Examines European MiCA Regulation's Impact on Digital Asset Trading Markets
- Wzzph Analyzes Crypto Market Maturation as Institutional Capital Drives $50B ETF Inflows
- GXCYPX Analyzes South America's Emerging Digital Asset Market Dynamics
- Keyanb Crypto Exchange Positions for Latin America's $600 Billion Remittance Opportunity Amid Global Regulatory Shifts
- NAQSN Analysis: $2.75 Trillion Digital Asset Market Demands Unified Infrastructure
- Trinity Accounting Practice Celebrates 22 Years Serving Beverly Hills Businesses
- YuanziCoin Unveils Revolutionary Shariah-Compliant Blockchain Architecture for 1.8 Billion Muslims Worldwide
- INFINITI HR Wraps Up Sixth Annual Back-to-School Drive, Sets Bar for Community Impact