Trending...
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- Veteran Launches GTG Energy: Nicotine-Free Pouch as Americans Rethink Addiction, Focus, and What Fuels Performance
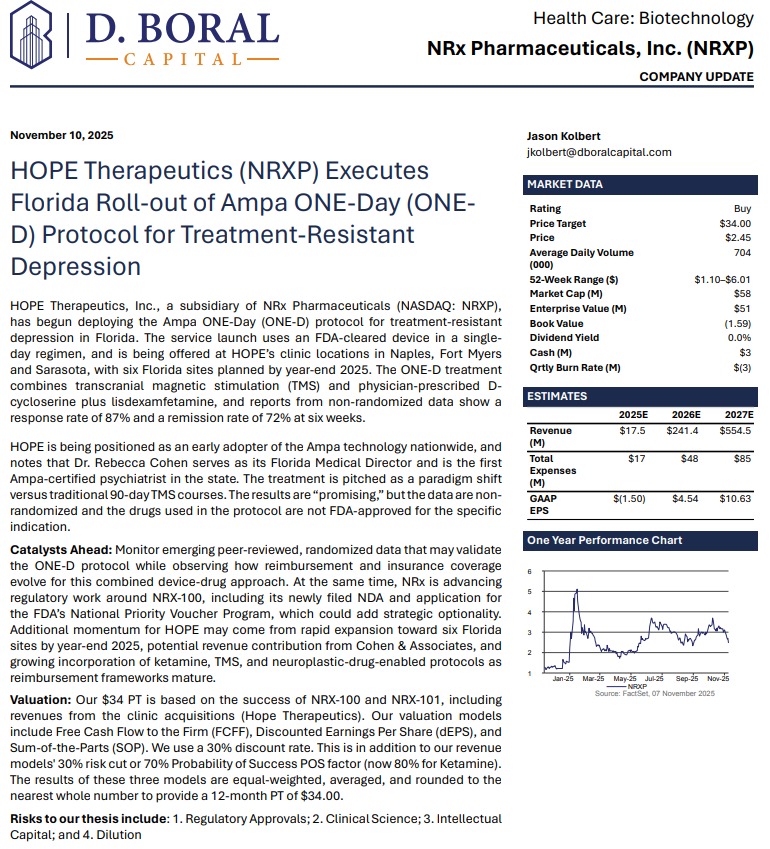
H.C. Wainwright Initiates Coverage with $34 Price Target, Citing Paradigm Shift in Depression Treatment. NRx Pharmaceuticals (N A S D A Q: NRXP) $NRXP
MIAMI - Marylandian -- NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP), a clinical-stage biopharmaceutical company pioneering NMDA-based therapeutics for central nervous system disorders, is emerging as a potential leader in the next generation of depression and suicidality treatments. With its innovative ONE-D (One Day Depression) protocol now launched in Florida and a $40 price target set by H.C. Wainwright, NRXP appears positioned for transformational growth in a rapidly expanding $3.35 billion global market.
A Major Step Forward: ONE-D Depression Treatment Launches in Florida
NRx Pharmaceuticals has initiated the first-in-Florida rollout of the ONE-D (One Day) Depression Treatment, in partnership with Ampa Health. The innovative protocol has shown breakthrough potential in achieving remission from treatment-resistant depression within a single day—an unprecedented leap forward for millions suffering from chronic depression.
Unlike traditional transcranial magnetic stimulation (TMS) therapies requiring up to 90 sessions over three months, the ONE-D approach combines a single-day TMS treatment with physician-prescribed compounds, including D-cycloserine, an active component of NRx's investigational NRX-101, and lisdexamfetamine. Early clinical data show an 87% response rate and 72% remission rate in nonrandomized studies.
The Ampa device is now available across NRXP HOPE Clinics in Sarasota, Naples, and Fort Myers, Florida—with six total locations expected by the end of 2025. This initiative marks one of the first national deployments of the Ampa ONE-D protocol.
More on Marylandian
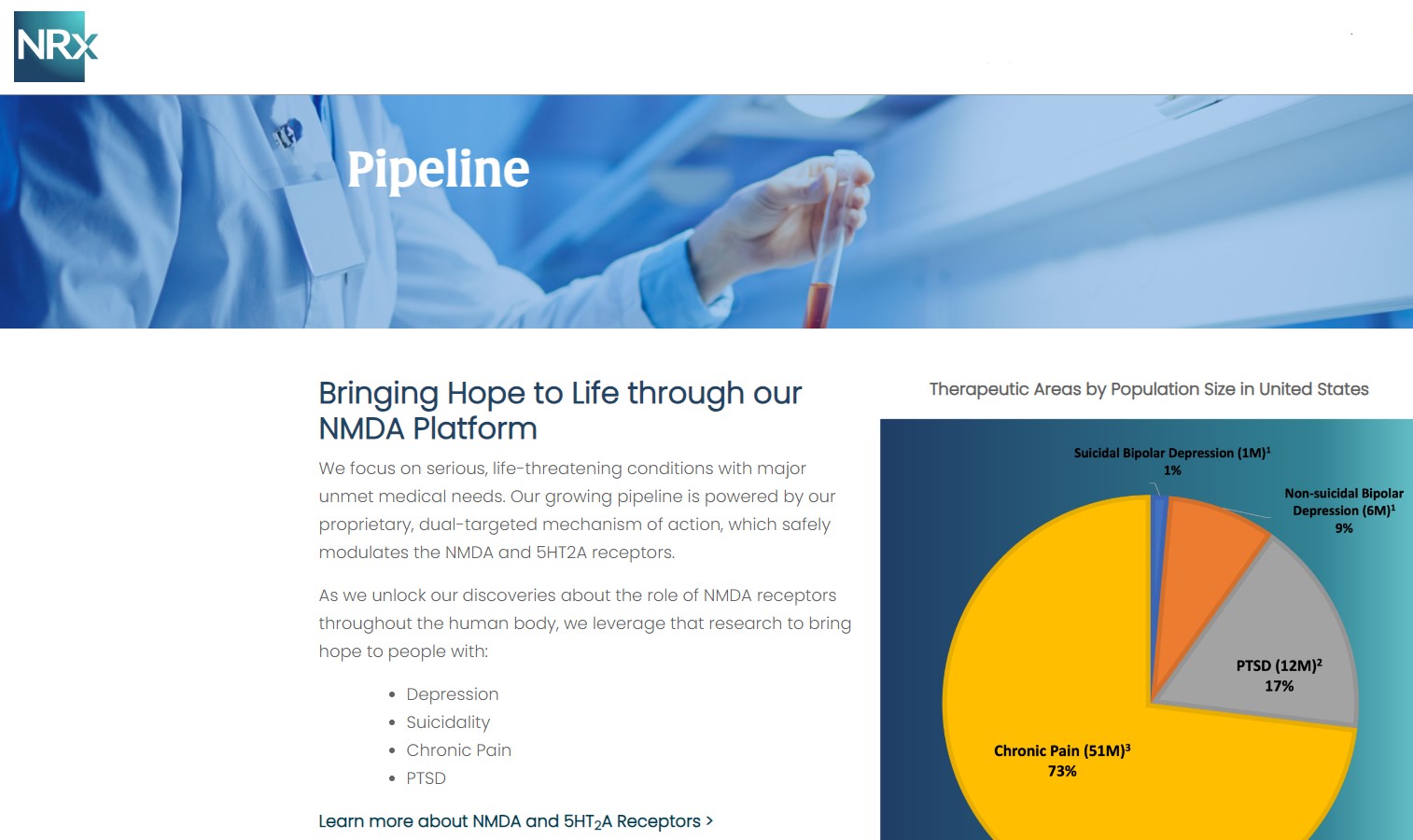
Breakthrough Therapeutic Portfolio: NRX-101 and KETAFREE™
At the center of NRXP's drug pipeline is NRX-101, an FDA-designated Breakthrough Therapy targeting suicidal, treatment-resistant bipolar depression and chronic pain. The drug represents a potentially lifesaving option for the more than 13 million Americans who seriously consider suicide each year, according to the CDC.
NRXP is also advancing KETAFREE™, a preservative-free IV ketamine formulation, through a refiled Abbreviated New Drug Application (ANDA) with the FDA. This comes as the ketamine market—currently valued at $750 million—is projected to reach $3.35 billion globally by 2034.
Importantly, the FDA has granted approval of NRXP's Suitability Petition for its proposed strength of preservative-free ketamine, clearing the path for potential market entry amid ongoing national shortages of sterile ketamine formulations.
Strategic Expansion Through Dura Medical Acquisition
NRXP recently completed its acquisition of Dura Medical, a revenue-generating and EBITDA-positive network of precision psychiatry clinics along Florida's West Coast. The addition of Dura, alongside pending acquisitions such as Neurospa TMS and Cohen & Associates, positions NRXP to deliver an integrated continuum of mental health care spanning TMS, ketamine therapy, and advanced pharmacologic treatments for PTSD, depression, and chronic pain.
Robust Financial and Partnership Outlook
The company's growth strategy is supported by a $7.8 million debt financing deal with Universal Capital, LLC, aimed at expanding NRXP's HOPE Clinic network. In parallel, NRXP has accepted non-binding potential licensing terms for NRX-100, with the agreement representing over $300 million in milestone payments and tiered double-digit royalties—a major value inflection opportunity for investors.
More on Marylandian
Moreover, NRXP's partnership with Alvogen Pharmaceuticals supports the ongoing development and commercial readiness of NRX-101, further validating the company's clinical and commercial trajectory.
Analyst Confidence: H.C. Wainwright Issues "Buy" Rating and $40 Price Target
A recent analyst report from H.C. Wainwright & Co. titled "A Paradigm Shift in the Treatment of Depression with Suicidality" initiated coverage on NRx Pharmaceuticals with a Buy rating and a $34 price target. The report underscores NRXP's leadership in transforming how depression and suicidality are treated, leveraging FDA Fast Track and Breakthrough Therapy designations to accelerate commercialization.
"We view NRXP's approach as a paradigm shift in the treatment of depression with suicidality," wrote H.C. Wainwright analysts. "With novel drug candidates, a rapidly expanding clinic network, and strategic licensing opportunities, NRXP is uniquely positioned to capture substantial market share."
About NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
NRx Pharmaceuticals, Inc. is a clinical-stage biopharmaceutical company developing breakthrough therapeutics based on its NMDA receptor modulation platform for the treatment of central nervous system disorders, including suicidal bipolar depression, chronic pain, and PTSD.
Through its partnerships, innovative clinical pipeline, and integrated treatment model, NRXP is setting new standards in precision psychiatry and interventional mental health care.
For More Information:
Company: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com
Website: www.nrxpharma.com
Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
A Major Step Forward: ONE-D Depression Treatment Launches in Florida
NRx Pharmaceuticals has initiated the first-in-Florida rollout of the ONE-D (One Day) Depression Treatment, in partnership with Ampa Health. The innovative protocol has shown breakthrough potential in achieving remission from treatment-resistant depression within a single day—an unprecedented leap forward for millions suffering from chronic depression.
Unlike traditional transcranial magnetic stimulation (TMS) therapies requiring up to 90 sessions over three months, the ONE-D approach combines a single-day TMS treatment with physician-prescribed compounds, including D-cycloserine, an active component of NRx's investigational NRX-101, and lisdexamfetamine. Early clinical data show an 87% response rate and 72% remission rate in nonrandomized studies.
The Ampa device is now available across NRXP HOPE Clinics in Sarasota, Naples, and Fort Myers, Florida—with six total locations expected by the end of 2025. This initiative marks one of the first national deployments of the Ampa ONE-D protocol.
More on Marylandian
- Event Solutions Enters New Era: Announces New Leadership
- Carlsbad Hotel Named Best of La Quinta Award Winner
- Maryland: NEWS RELEASE: Caroline County Control Area Released
- Maryland Agricultural Fair Board Meeting Notice
- Scoop Social Co. Launches a New Era of Mobile Hospitality — One Truck, Two Experiences
Breakthrough Therapeutic Portfolio: NRX-101 and KETAFREE™
At the center of NRXP's drug pipeline is NRX-101, an FDA-designated Breakthrough Therapy targeting suicidal, treatment-resistant bipolar depression and chronic pain. The drug represents a potentially lifesaving option for the more than 13 million Americans who seriously consider suicide each year, according to the CDC.
NRXP is also advancing KETAFREE™, a preservative-free IV ketamine formulation, through a refiled Abbreviated New Drug Application (ANDA) with the FDA. This comes as the ketamine market—currently valued at $750 million—is projected to reach $3.35 billion globally by 2034.
Importantly, the FDA has granted approval of NRXP's Suitability Petition for its proposed strength of preservative-free ketamine, clearing the path for potential market entry amid ongoing national shortages of sterile ketamine formulations.
Strategic Expansion Through Dura Medical Acquisition
NRXP recently completed its acquisition of Dura Medical, a revenue-generating and EBITDA-positive network of precision psychiatry clinics along Florida's West Coast. The addition of Dura, alongside pending acquisitions such as Neurospa TMS and Cohen & Associates, positions NRXP to deliver an integrated continuum of mental health care spanning TMS, ketamine therapy, and advanced pharmacologic treatments for PTSD, depression, and chronic pain.
Robust Financial and Partnership Outlook
The company's growth strategy is supported by a $7.8 million debt financing deal with Universal Capital, LLC, aimed at expanding NRXP's HOPE Clinic network. In parallel, NRXP has accepted non-binding potential licensing terms for NRX-100, with the agreement representing over $300 million in milestone payments and tiered double-digit royalties—a major value inflection opportunity for investors.
More on Marylandian
- ADS and Gamma Scientific Release Joint White Paper on Practical Path to NHTSA ADB Compliance
- Record Sales Growth After Strategic Acquisitions; New Distribution Agreements for Established Premium Cigar Supplier: Green Leaf Innovations $GRLF
- R2 Copilot Addresses Critical Privacy Issues as Enterprise AI Spending and Security Incidents Rise
- Innovative Environmental Technologies Unveils New Website Featuring Free AI Tools for the Environmental Industry
- CCHR Warns: Psychiatric Diagnoses Without Biological Proof Now Used to Justify Euthanasia
Moreover, NRXP's partnership with Alvogen Pharmaceuticals supports the ongoing development and commercial readiness of NRX-101, further validating the company's clinical and commercial trajectory.
Analyst Confidence: H.C. Wainwright Issues "Buy" Rating and $40 Price Target
A recent analyst report from H.C. Wainwright & Co. titled "A Paradigm Shift in the Treatment of Depression with Suicidality" initiated coverage on NRx Pharmaceuticals with a Buy rating and a $34 price target. The report underscores NRXP's leadership in transforming how depression and suicidality are treated, leveraging FDA Fast Track and Breakthrough Therapy designations to accelerate commercialization.
"We view NRXP's approach as a paradigm shift in the treatment of depression with suicidality," wrote H.C. Wainwright analysts. "With novel drug candidates, a rapidly expanding clinic network, and strategic licensing opportunities, NRXP is uniquely positioned to capture substantial market share."
About NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
NRx Pharmaceuticals, Inc. is a clinical-stage biopharmaceutical company developing breakthrough therapeutics based on its NMDA receptor modulation platform for the treatment of central nervous system disorders, including suicidal bipolar depression, chronic pain, and PTSD.
Through its partnerships, innovative clinical pipeline, and integrated treatment model, NRXP is setting new standards in precision psychiatry and interventional mental health care.
For More Information:
Company: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com
Website: www.nrxpharma.com
Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on Marylandian
- MSTRO Introduces Motif™ Marketplace, Turning mSpace into a Streaming AI Application Ecosystem
- MWA Prince George's County Hosts "From Shy to Sold" with Publishing Consultant Ally Machate
- Training Lofts Launches $1,099 Unlimited Training Membership Featuring Semi-Private Coaching, Nutrition Support, and Recovery Services
- Maryland: NEWS RELEASE: Preliminary Testing Confirms Highly Pathogenic Avian Influenza in Cecil County
- Deliverance Ministry Ordination Training Announced on Zoom
- DMV Families Choose Regional Staycations Over Long-Distance Travel at Frederick County's
- American Properties Realty, Inc. Leadership Attends NAHB International Builders' Show in Florida
- Women's Personal Trainer Stephen Holt Named Finalist for IDEA Personal Trainer of the Year
- Exploring Kunta: Roots 50 Commemorative Experience
- $317M Revenue and a Clear Path to $1B: $IQST is Positioned for a Major Profitability Inflection
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- Scotch Whisky Market Dislocation Creates Compelling Entry Opportunity for Long-Term Investors
- Peccioli Becomes New Orleans: In July 2026, the magic of jazz comes to Tuscany
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- The "Unsexy" Business Quietly Creating 130+ New Entrepreneurs Across America — From Alaska to Puerto Rico
- Veteran Launches GTG Energy: Nicotine-Free Pouch as Americans Rethink Addiction, Focus, and What Fuels Performance
- RecallSentry™ App Launch — Your Home Safety Hub — Free on iOS & Android
- Award-Winning Director Crystal J. Huang's Under-$50K Film "The Ritual House" Wins Best Horror Feature at Golden State Film Festival